By: Dana Sackett
Consuming too many nutrients is something many of us can understand following the holidays. Similar to ourselves too many nutrients in streams, rivers, lakes, and oceans can cause major health problems. In fact, excess nutrients that enrich plant growth in aquatic ecosystems has a special name: eutrophication. While this term was originally used to describe the natural aging process of a lake (where a lake will over time become more nutrient rich; eventually filling in to become a pond and then a marsh) it is now most often used to describe the accelerated input (by hundreds or thousands of years) of nutrients into lakes, rivers and oceans created by human activities.

When scientists talk about nutrients in aquatic systems, we are mostly talking about nitrogen and phosphorus, two of the basic components needed to feed primary producers, or the plants and algae (also referred to as phytoplankton) that create oxygen and make up the base of foodwebs. Some of the most common sources of nutrients in water are agricultural runoff from fertilizers and livestock waste, wastewater treatment plants, aquaculture, waste from fish and other animals that live in or near the water, and atmospheric deposition. Of these sources some have a much bigger impact than others.

Industrial and agricultural developments over the last several decades have led to a drastic increase in nutrient input into aquatic ecosystems. This increase has altered the nutrient cycle in ground and surface waters, changed the acidity of water, and deteriorated water quality around the globe. For example, nitrate, a form of nitrogen, is presently one of the most common contaminants in groundwater and has been linked to ‘blue baby syndrome’ which decreases the ability of blood to carry oxygen throughout the body, most commonly affecting infants. Indeed, excess nutrients have been quoted by some scientists to be the Earth’s most widespread water quality problem.

The excessive input of nutrients into our waters has also caused rapid increases in populations of harmful toxin-producing algae (toxic algal blooms) that have lead to fish kills and human illness. Algae blooms can also be harmful through decomposition. The decomposition of algae requires the use of oxygen. When a large population of algae (a bloom) is formed, it dies shortly thereafter and is decomposed by bacteria. The bacteria eat up all the oxygen in the water, suffocating the aquatic life in the area. As a result, these areas are sometimes referred to as dead zones. These blooms and dead zones are common near large human populations and have been increasing in frequency over the last several decades.

Scientists are hard at work trying to find solutions to the problem of eutrophication. Some have found ways to trace nitrogen and phosphorus in water using isotopes to better understand how these nutrients cycle and where they originate. Others have suggested methods to reduce nutrient inputs from wastewater treatment plants using more sophisticated equipment and the reduction of fertilizer-use by agriculture; while still others have discovered the exact ratios and types of nutrients that lead to blooms of specific toxic algal species, and how all of these nutrients are changing aquatic foodwebs. Several laws and regulations limit nitrogen and phosphorus inputs in the United States and elsewhere. Continued research helps to evaluate whether these rules are sufficient to maintain water quality standards.
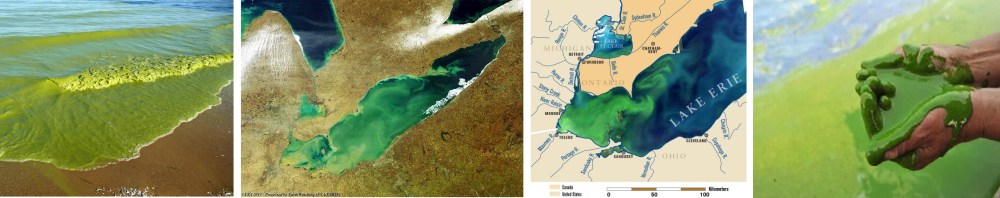
Although the problem of excess nutrients is widespread, there are also places that have little, if any, nutrient inputs (these places are called oligotrophic). Have you ever admired the crystal clear blue water in a tropical paradise? If so, you were probably enjoying nutrient poor water. Indeed, nutrient levels in the ocean can often be inferred by water color from space. Where there is plenty of food, there are often plenty of algae to eat that food. Algae absorb certain wave-lengths of light for energy, blue light being one of them, leaving behind green wave-lengths of light to be reflected off the ocean, making the water look green. Conversely, if there are little to no nutrients or algae the blue light is not being absorbed and the ocean looks blue.

There is a great deal more information on nutrients in water and eutrophication then what has been presented here. If you are interested in learning more please check out some of the articles and links below.
References and additional information:
Anderson DM, Gilbert PM, Burkholder JM. 2002. Harmful algal blooms and eutrophication: nutrient sources, composition, and consequences. Estuaries 25: 704-726.
Cai W, Hu X, Huang W, Murrell MC, Lehrter JC, Lohrenz SE, Chou W, Zhai W, Hollibaugh JT, Wang Y, Zhao P, Guo X, Gundersen K, Dai M, Gong G. 2011. Acidification of subsurface coastal waters enhanced by eutrophication. Nature Geoscience. DOI: 10.1038/NGEO1297
Correll DL. 1998. The role of phosphorus in the eutrophication of receiving waters: a review. Journal of Environmental Quality 27:261-266.
Deegan LA, Johnson DS, Warren RS, Peterson BJ, Fleeger JW, Fagherazzi S, Wollheim WM. 2012. Coastal eutrophication as a driver of salt march loss. Nature 490:388-394.
Howarth R, Chan F, Conley DJ, Garnier J, Doney SC, Marino R, Billen G. 2011. Coupled biogeochemical cycles: eutrophication and hypoxia in temperate estuaries and coastal marine ecosystems. Frontiers in Ecology and the Environment 9:18-26.
Layman CA, Allgeier JE, Yeager LA, Stoner EW. 2013. Thresholds of ecosystem response to nutrient enrichment from fish aggregations. Ecology 94:530-536.
Meador MR. 2013. Nutrient enrichment and fish nutrient tolerance: assessing biologically relevant nutrient criteria. Journal of the American Water Resources Association 49:253-263.
Mueller ND, Gerber JS, Johnston M, Ray DK, Ramankutty N, Foley JA. 2012. Closing yield gaps through nutrient and water management. Nature 490:254-257.
Ryther JH, Dunstan WM. 1971. Phosphorus, and eutrophication in the coastal marine environment. Science 171:1008-1013.
Smith SV, Swaney DP, Buddemeier RW, Scarsbrook MR, Weatherhead MA, Humborg C, Eriksson H, Hannerz F. 2005. River nutrient loads and catchment size. Biogeochemistry 75:83-107.
http://www.lakescientist.com/lake-facts/water-quality/
http://science.nasa.gov/earth-science/oceanography/living-ocean/ocean-color/
http://blog.nwf.org/2014/05/protecting-wildlife-from-freshwater-harmful-algal-blooms/
http://www.who.int/water_sanitation_health/diseases/methaemoglob/en/
http://earthsky.org/earth/2011-gulf-of-mexico-dead-zone-smaller-than-scientists-predicted
http://www.upi.com/Science_News/2013/04/09/Agriculture-blamed-in-ocean-Dead-Zone/59221365545765/
