By: Dana Sackett
You have probably heard the myth that baby snakes are more dangerous than adults because they haven’t learned to control the amount of venom they inject. The theory being that baby snakes unintentionally release much more than an adult would when they bite. While this myth has been mostly dispelled by herpetologists (see article here) there is something to the notion that young and adult animal venom can differ. For some organisms, rather than the amount released, it’s the actual recipe of the venom that can change. A surprising discovery being that scientists previously assumed the venom for a single species or individual remained the same throughout their lifetime.

The compounds that make-up venom can vary widely across organisms, with each type seemingly specific to a particular target and function. For example, defensive venoms (which many fish have) are often highly conserved with the primary action being immediate extreme localized pain. Conversely, the recipe for predatory venoms (used to catch prey) are often more complex and variable and affect other animals in different ways. To make matters even more complicated, in addition to the expected differences among species, there are also differences in venom among closely-related species, within the same species, within the same family (sex-related differences in siblings), and even within the same individual over its lifetime.

For instance, sea anemones, famous from Disney’s “Finding Nemo” as Nemo’s home, have stinging cells on their tentacles that inject venom into the animals they touch. Very recently researchers discovered that the toxins expressed in each of the complex life cycle stages of the sea anemone (egg, to barely visible swimming larvae, to metamorphosed sessile polyp, to mature adult) were different and specific to the predators they were most vulnerable to or the prey they were targeting in each life stage. This means that the composition of the venom for these individuals changed from a defensive venom to a predatory venom over their life time.

Australian box jellyfish (see here to read more about these very venomous creatures) demonstrated that their venom changed from when they were immature targeting invertebrate prey to when they were mature targeting larger vertebrate prey. For one cone snail species (another group of very venomous creatures), an individual produced two distinctly different venoms over the course of a few months, a change that was unrelated to age or size. In another study, individual cone snails of another species produced distinctly different venoms in response to either defensive or predatory stimuli. In fact, the predatory venom from those individuals were specific to their prey and harmless to humans. However, the defense-evoked venom contained high levels of paralytic toxins consistent with lethal effects on humans.
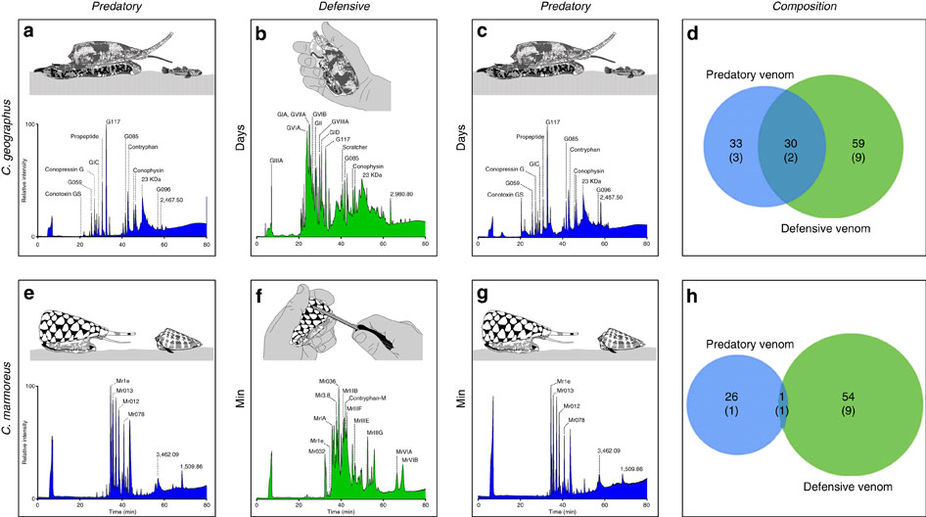
Nature is remarkable in its intricacies and nuances. While the study of venomous creatures helps to understand the interactions between predators and prey and how those interactions have evolved over time, the result of such variation can have big consequences on the effectiveness of anti-venoms. Thus, knowing how, why, and where these variations exists could help with the development of more effective anti-venoms, medicines, and in understanding the fascinating processes responsible for producing some of the most complex and potent biochemical secretions found on the globe.
References and additional reading material
Casewell NR, Wuster W, Vonk FJ, Harrison RA, Fry BG. 2013. Complex cocktails: the evolutionary novelty of venoms. Trends in Ecology and Evolution 28: 219-229.
Dutertre S, Biass D, Stocklin R, Favreau P. 2010. Dramatic intraspecimen variations within the injected venom of Conus consors: an unsuspected contribution to venom diversity. Toxicon 55: 1453-1462.
Dutertre S, Jin A, Vetter I, Hamilton B, Sunagar K, Lavergne V, Dutertre V, Fry BG, Antunes A, Venter DJ, Alewood PF, Lewis RJ. 2014. Evolution of separate predation- and defence-evoked venoms in carnivorous cone snails. Nature Communications 5:3521 DOI: 10.1038/ncomms4521
Ernst CH 1992. Venomous Reptiles of North America. Smithsonian Institution Press, Washington, D.C.
Jester R, Lefebvre K, Langlois G, Vigilant V, Baugh K, Silver MW. 2009. A shift in the dominant toxin-producing algal species in central California alters phycotoxins in food webs. Harmful Agae 8: 291-298.
Li M, Fry BG, Kini RM. 2005. Eggs-only diet: its implications for the toxin profile changes and ecology of marbled sea snake (Aipysurus eydouxii). Journal of Molecular Evolution 60: 81-89.
Mehrtens, John M. 1987. Living Snakes of the World in Color. Sterling Press, New York, New York.
Safavi-Hemami H, Siero WA, Kuang Z, Williamson NA, Karas JA, Page LR, MacMillan D, Callaghan B, Kompella SN, Adams DJ, Norton RS, Purcell AW. 2011. Embryonic toxin expression in the cone snail Conus victoriae: primed to kill or divergent function? The Journal of Biological Chemistry 286: 22546-22557.
Underwood AH, Seymour JE. 2007. Venom ontogeny, diet and morphology in Carukia barnesi, a species of Australian box jellyfish that causes Irukandji syndrome. Toxicon 49: 1073-1082.
https://www.sciencedaily.com/releases/2018/03/180305101633.htm
http://www.reptilesmagazine.com/Snake-Myths-and-Facts/
https://www.sciencenews.org/article/venomous-fish-have-evolved-many-ways-inflict-pain

Very good article here. I am very happy to read this post and looking to see more good post about this post. Thank you very much for sharing this post. Carry on it. Very well website it is. I like the blog you share on this website the content of your website is very good. Thanks for sharing this. I must share this website to get more update and also recommend this with others. I am satisfied to peer your post. Thank you so much and I am looking ahead to touch you. Keep sharing.