By: Dana Sackett, PhD
Recent research has found that fish may be more important to the ocean carbon cycle than previously predicted. A compound called calcium carbonate, which is the main ingredient in the shells of marine organisms (coral reefs, oysters, clams, sea urchins, some plankton), is predicted to become more and more limited to these organisms as a result of climate change. But how do fish fit into this picture and how could they help to balance the lowered available calcium carbonate to these organisms. To answer this question, first we need to briefly look at how increasing carbon dioxide in the air impacts this important building block in the ocean.

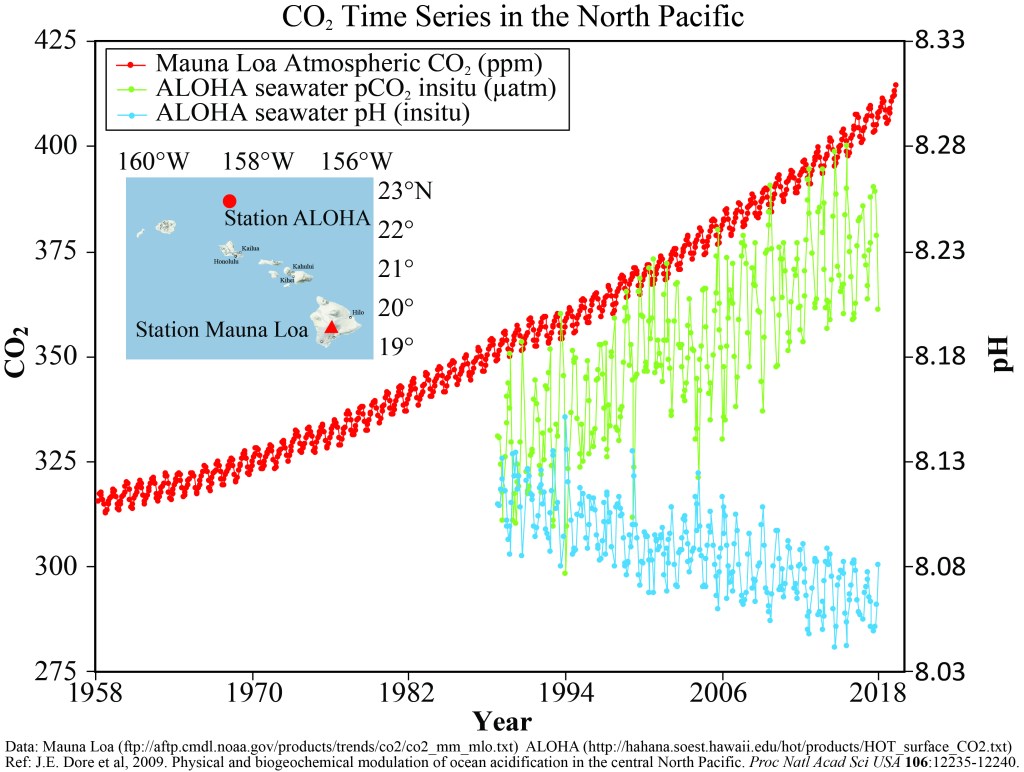
Most gases such as oxygen tend to be in the atmosphere rather than in the ocean. For instance, more than 99% of the gas form of oxygen is in the atmosphere, leaving very little by comparison dissolved in the ocean. Carbon dioxide on the other hand does not follow this general rule. For instance, scientist have estimated that more than 98% of the carbon dioxide that was released before the industrial revolution is in the ocean and only about 1.5% of that is in the atmosphere. The reason is that carbon dioxide can dissolve much more easily in seawater, and because when it does dissolve, the water (H2O) uses the carbon dioxide to make new compounds with extra hydrogen ions as a byproduct of this process. Increased hydrogen ions make any liquid more acidic. In fact, the definition of an acid is a liquid with excess hydrogen ions. These hydrogen ions steal the carbonate that would otherwise be used to make calcium carbonate, to make a different compound instead. Therefore, the more carbon dioxide in the atmosphere the more it will dissolve in the ocean and the more hydrogen ions will steal carbonate away from marine organisms (this process is called ocean acidification).
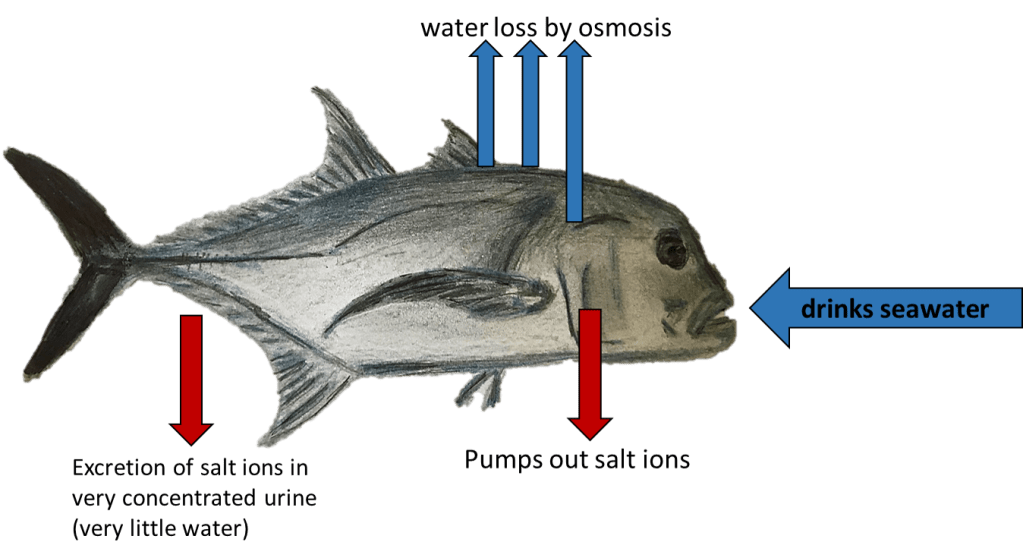
So how can a fish help in this situation? The answer lies with osmosis. Osmosis is a process that most people first learn about in school. This process occurs when water flows across a semi-permeable barrier (meaning some things can pass through it) from an area of low concentration to an area of high concentration in an effort to balance the concentration in the two areas. Osmosis poses a significant issue for fish because it is constantly trying to balance the internal fluids of fish with their surrounding environment. This means that for freshwater fish, which are saltier than the surrounding water, the freshwater around them is constantly passing into their body to dilute them to match the outside environment. On the other hand, saltwater fish are constantly losing water from their tissues as osmosis tries to make their internal fluids just as salty as the saltier water around them. The methods and energy fish use to prevent their bodies from either bursting with water or shriveling up like a raisin is called osmoregulation.

It is because of this process that fish have been found to help out with the carbonate being stolen from those shelled marine organisms. Marine fish are constantly drinking seawater to replace the water continuously being lost from their bodies (ever wonder where the phrase drink like a fish came from, now you know). However, seawater is too salty so these fish use energy to kick-out all of the excess salts and minerals in the seawater they ingest. This means that marine fish are constantly kicking calcium carbonate out of their bodies and into the ocean in an effort to get rid of excess calcium. While this process has been well known by scientists for a while it had not been considered in models of ocean chemistry until very recently.
The models created by a recent study conservatively estimated that bony fish produce 40 million–110 million tonnes of calcium carbonate per year. That is a lot of calcium carbonate that excesses hydrogen ions can steal from, that don’t come from shelled marine organisms. Scientists also believe this previously unconsidered source of calcium carbonate might be why ocean surface waters are a bit less acidic than models have predicted. In addition, a scientist from NOAA has suggested that the inclusion of the calcium carbonate produced from fish may be the answer to a dilemma scientists were having with the calcium-carbonate budget in the ocean and thus, a key to balancing the marine carbon cycle.
References and other reading material:
Kwok R. 2009. Fish are crucial in oceanic carbon cycle. Nature: naturenews. doi:10.1038/news.2009.30
Marinov I., Sarmiento J.L. (2004) The Role of the Oceans in the Global Carbon Cycle: An Overview. In: Follows M., Oguz T. (eds) The Ocean Carbon Cycle and Climate. NATO Science Series (Series IV: Earth and Environmental Sciences), vol 40. Springer, Dordrecht
Tanhua T, Bates NR, Kortzinger A. 2013. Chapter 30 – The marine carbon cycle and ocean carbon inventories. Internal Geophysics 103:787-815.
Wilson RW, Millero FJ, Taylor JR, Walsh PJ, Christensen V, Jennings S, Grosell M. 2009. Contribution of fish to the marine inorganic carbon cycle. Science 323: 359-362.
